Max Access Solutions is our innovative, patient-centered model for delivering cancer treatment to those in need. Through the program, launched in 2017, we connect people facing cancer in countries where treatment is not locally available to high-impact medicines, qualified medical professionals, and essential support services.
This work is made possible through partnerships with local physicians, ministries of health, other non-governmental organizations, patient organizations, and medicine manufacturers. Our partner physicians play a vital role in receiving the medication at the local level, enrolling patients into the program, and providing the individual care each patient needs.
Max Access Solutions is meant to bridge patients’ access to treatment in underserved areas, with a focus on low- and middle-income countries, until local access channels can be put in place.
- 35K+
patients provided access to medication and/or support services each year
- 80+
countries where medication access and/or support services are provided
- 15
medicines provided
Our Cancer Programs
For over 25 years, we’ve developed our cancer programs and expanded our partnerships to reach cancer patients in more than 80 low- and middle-income countries. We now offer a wide portfolio of life-extending medications for multiple disease areas and growing.

“The access to drugs, at no cost, has not only helped me financially but also enhanced my overall well-being. This has enabled me to sustain my occupation as a farmer, which demands immense endurance and physical strength. As a patient facing financial constraints, the provision of such vital medication is invaluable, and I cannot express enough gratitude to The Max Foundation, [the pharmaceutical company], and my treating physicians for facilitating access to this treatment.”
– Serali, a farmer, father, and grandfather from Nepal living with chronic myeloid leukemia
Latest Developments

CancerWorld Special Issue
We’re excited to announce our collaboration with CancerWorld magazine! This special issue is about the 25th anniversary of an oral medication for chronic myeloid leukemia that transformed treatment, diagnostics, access, and patient advocacy. It features articles from the global community, including clinicians, researchers, patients, and advocates. Learn more about how a woman living with CML….

Partner physician article published in Clinical Surgical Oncology
An article by one of our partner physicians, Dr. Freddy Gnangnon in Benin, was recently published in Clinical Surgical Oncology. It’s about the management of locally advanced gastrointestinal stromal tumors and how providing access to treatment free of charge improves survival outcomes. Read More

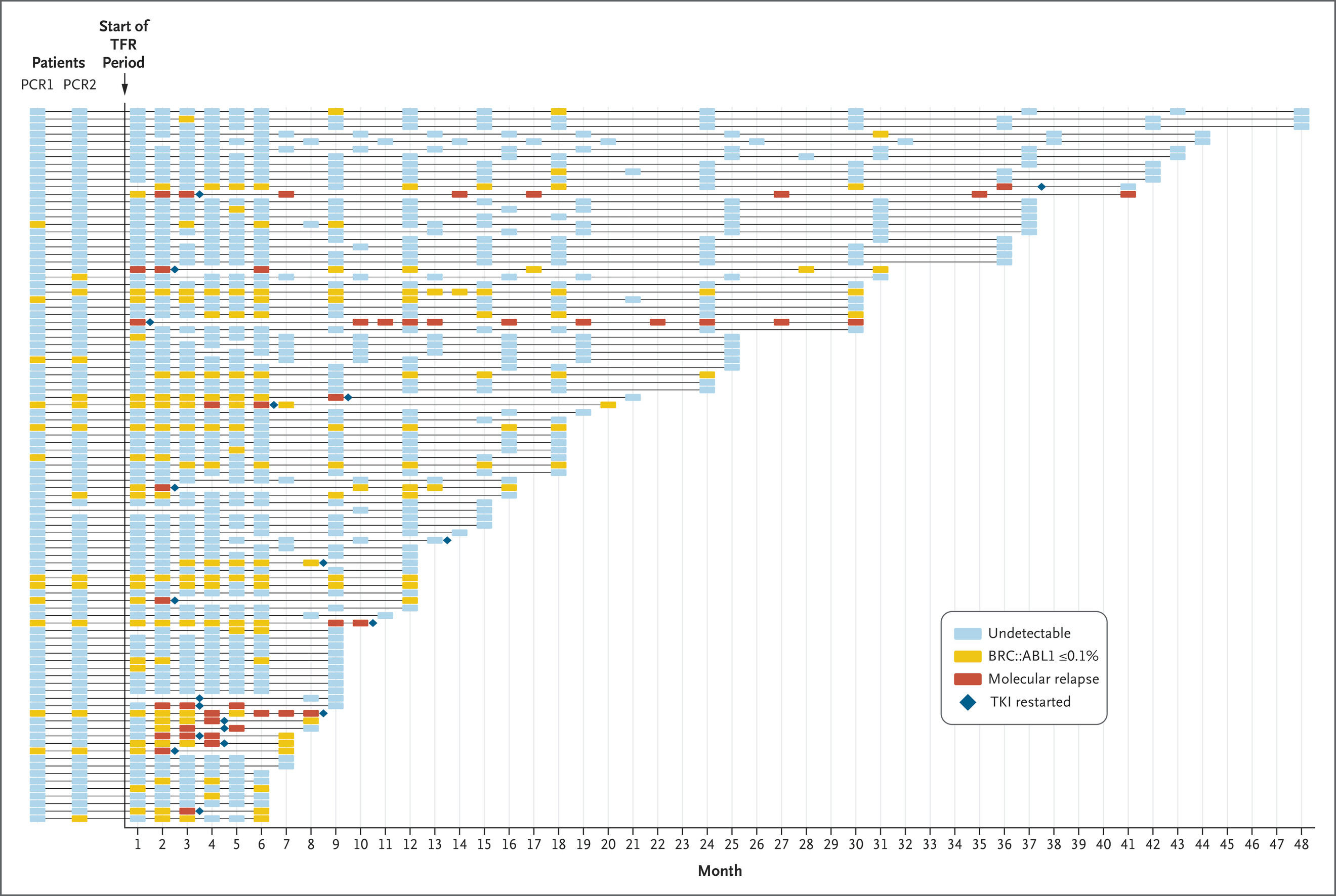
Treatment-Free Remission in Chronic Myeloid Leukemia in Low- and Middle-Income Countries
We’re pleased to share that the New England Journal of Medicine has published our “Letter to the Editor” on treatment-free remission for chronic myeloid leukemia in low- and middle-income countries! It was written in collaboration with Dr. Jerry Radich from the Fred Hutchinson Cancer Research Center and partner physicians from Armenia, the Dominican Republic, El….

Overcoming Barriers to Patient Access
Improving health outcomes for people living with cancer is about more than medication. We provide patient access solutions through psychosocial support, transportation assistance, adherence monitoring, and more to ensure patient needs are handled individually and holistically.
Learn More